 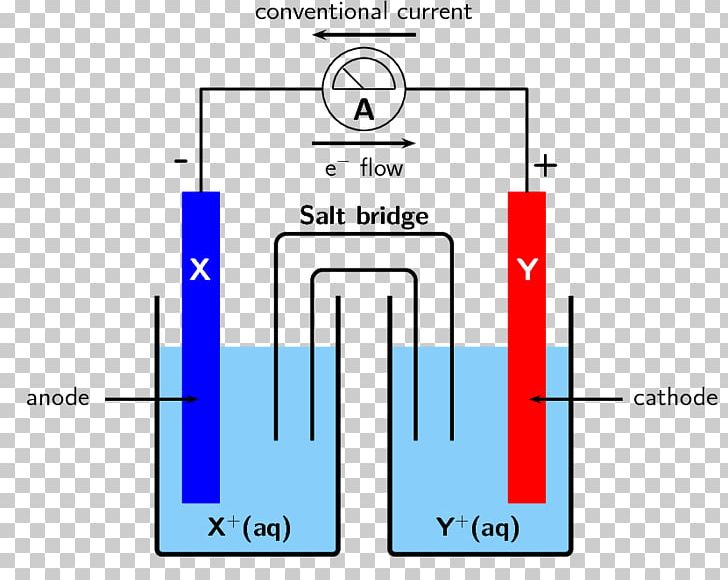
Accordingly, electrochemical cells used in batteries usually don't have a salt bridge but a semipermeable membrane instead, or a clever choice of half reactions where mixing of the electrolytes is not a problem (lead acid battery for example, where the only soluble species, sulfuric acid, is common to both half reactions). This tells you that the salt bridge will be depleted after a certain amount of current flows through the wire. To conclude, ions flow out of the salt bridge into the solutions A and B, but virtually no ion flows all the way through the salt bridge. The solution in the salt bridge is not mixed, so it would take a very long time for the electrolyte in A to diffuse all the way to B. When you have a high concentration of inert ions in the salt bridge, cations in the salt bridge will flow into B, and anions in the salt bridge will flow into A. The idea of the salt bridge is to prevent electrolytes mixing while providing ion flow. In the reaction, the silver ion is reduced by gaining an electron, and solid Ag is the cathode.According to my notes and many sources on the internet, electrons and cations both travel from the anode (A in the image) to the cathode (B in the image). In a typical voltaic cell, the redox pair is copper and zinc, represented in the following half-cell reactions: The silver is reduced at the cathode from Ag (I) to Ag, and the zinc is oxidized from Zn to Zn (II). This redox reaction consists of two half-reactions. A silver oxide battery uses silver (I) oxide as the positive electrode ( cathode ), zinc as the negative electrode ( anode ), plus an alkaline electrolyte, usually sodium hydroxide (NaOH) or potassium hydroxide (KOH). The operating principle of the voltaic cell is a simultaneous oxidation and reduction reaction, called a redox reaction. This flow of electrons is an electrical current that can be used to do work, such as turn a motor or power a light. The two electrodes must be electrically connected to each other, allowing for a flow of electrons that leave the metal of the anode and flow through this connection to the ions at the surface of the cathode. This forms a solid metal that deposits on the cathode. At the cathode, the metal ion in the solution will accept one or more electrons from the cathode, and the ion’s oxidation state will reduce to 0. The metal of the anode will oxidize, going from an oxidation state of 0 (in the solid form) to a positive oxidation state, and it will become an ion. The anode will undergo oxidation and the cathode will undergo reduction. The voltaic cell uses two different metal electrodes, each in an electrolyte solution. In between these electrodes is the electrolyte, which contains ions that can freely move. Electrodes can be made from any sufficiently conductive materials, such as metals, semiconductors, graphite, and even conductive polymers. The cathode is the electrode where reduction takes place. 
The anode is defined as the electrode where oxidation occurs. These scientists conducted several experiments on chemical reactions and electric current during the late 18th century.Įlectrochemical cells have two conductive electrodes, called the anode and the cathode. This kind of cell includes the galvanic, or voltaic, cell, named after Luigi Galvani and Alessandro Volta. voltaic cell: A cell, such as in a battery, in which an irreversible chemical reaction generates electricity a cell that cannot be recharged.Īn electrochemical cell is a device that produces an electric current from energy released by a spontaneous redox reaction.half-cell: Either of the two parts of an electrochemical cell containing an electrode and an electrolyte.redox: A reversible chemical reaction in which one reaction is an oxidation and the reverse is a reduction.The half-cells are connected by a salt bridge that allows the ions in the solution to move from one half-cell to the other, so that the reaction can continue.The electrons always flow from the anode to the cathode.Reduction describes the gain of electrons by a molecule, atom, or ion.Oxidation describes the loss of electrons by a molecule, atom, or ion. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
